Crinetics is a pharmaceutical company that develops much-needed therapies for people with rare endocrine diseases. We’re here for patients who are eager to find therapies that provide effective disease control and more simplicity in their lives. We partner with healthcare practitioners to ensure we’re solving real problems for them. And we build value in the company for investors by filling real market needs. Whatever brought you here, welcome.
An Entire Pipeline of Progress
Intellectual rigor, sound science, and innovative approaches are the hallmarks of our work and the way we maintain a healthy pipeline.
We focus on the discovery of drugs for rare endocrine disease and related tumors with:
- High unmet medical need
- Established biology
- Biomarker endpoints
- Early phase POC
- Efficient registration trials
| Discovery | Pre-Clinical | Phase 1 | Phase 2 | Phase 3 |
|---|---|---|---|---|
Acromegaly (PATHFNDR-1)
| Discovery | Pre-Clinical | Phase 1 | Phase 2 | Phase 3 |
|---|---|---|---|---|
Acromegaly (PATHFNDR-1)
Meet Our Top Team of Endocrinologists
Crinetics was founded in endocrinology research and we’ve expanded our ranks with scientists and doctors who are leading experts in the field.
“Endocrinology is multi-dimensional. You have to think about the body holistically, As a company, Crinetics shares my point of view that it’s one thing to know all the hormonal fluctuations and their implications, but it’s also important to know what those fluctuations mean to an individual patient and how we can help that person get better, given that knowledge.”

Peter Trainer, M.D., FRCPE, Vice President of Clinical Endocrinology
- Honorary professor of endocrinology, University of Manchester (UK)
- Clinical Director, Manchester Academic Health Science Centre
- Director, Royal College of Physicians of Edinburgh
- Associate Medical Director/Clinical Lead for endocrinology, The Christie NHS Foundation Trust
- Sr Exec Committee member: Society for Endocrinology, the Endocrine Society & European Society of Endocrinology (ESE)
“I’ve been involved with a number of companies developing products for endocrinology disorders during my career. Crinetics’ ethos of science-driven innovation aligns perfectly with my interest and experience as a clinical scientist and in driving the translation of bench-top discovery through the developmental steps necessary for deployment for patient benefit.”

Alessandra Casagrande, M.D., Ph.D.,Medical Sciences Director
- Clinical researcher on rare diseases, Universidade Federal de Sao Paulo, Brazil
- Researcher, Endocrinology,
Rush University Medical Center and University of Illinois, Chicago - Medical Director, Rare Disease, pharmaceutical company, Sao Paulo, Brazil
- Attending physician, Fleury Medicina Diagnóstica, Sao Paulo, Brazil
- Clinical trials sub-investigator, Hospital de Clinicas, Porto Alegre, Brazil
“Neuroendocrinology has traditionally been an underserved discipline in that it doesn’t get the level of research funding many other disciplines do. I like that Crinetics recognized this from the start and pours so much effort and energy into making up for that. Rare endocrine diseases can have cascading effects on patients’ health and lives. Contributing to the solutions is rewarding work.”

“Providing hope and options for people seeking therapies for rare diseases has been my passion for over 15 years. My experience with medicine and drug development is aligned with Crinetics’ remarkable insights and expertise in rare endocrine diseases. We share a long-term commitment to serving these patients and to developing life-saving products to meet their needs.”

Christine Ferrara-Cook, MD, PhDSenior Medical Director
- Director of Clinical Development, Rezolute
- External Clinical Consultant, pH PHARMA
- Assistant Professor, Pediatric Endocrinology, University of California, San Francisco
- Fellowship/Internship/Residency, Pediatric Endocrinology, The Children’s Hospital of Philadelphia
“What attracted me to Crinetics was the Discovery Kitchen, as I call it. We have an in-house Discovery team which is something so unique in a pharmaceutical company. It allows you the opportunity to find patients and diseases for which there is the most unmet need and really be committed to a patient population or a disease.”
Let's Get Acromegaly on Your Radar
Acromegaly is a very rare disorder that develops when a benign tumor grows on the pituitary gland and secretes increased levels of growth hormone (GH) during adulthood. It can develop at any age but usually affects middle-aged adults.
Prevalence
About 25,000 patients in the U.S.
Incidence rate
3,000 new cases per year
Median age at diagnosis
40.5 – 47 years
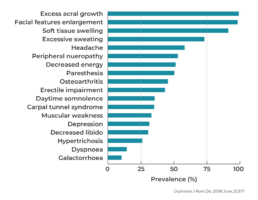
Clinical manifestations of acromegaly can vary. Common changes in facial appearance can include a larger nose, fuller lips, a protruding lower jaw and brow, more numerous skin folds, and a general appearance of being unwell. Loss of peripheral vision is also possible as the tumor compresses the optic chiasm. Patients may report newly developed difficulty parallel parking, a general fogginess or forgetfulness, and other symptoms, as seen in this chart.
Current medical therapies require painful monthly injections that disrupt patients’ lives, or oral medication that calls for a dietary regimen that can be difficult to maintain. Crinetics is developing a once-daily oral treatment for acromegaly called paltusotine that may offer patients a better option.
We Are Here For the Patients
To us, patients are more than the symptoms they have or the treatments they’re on. You are partners in this work. Real people with friends and families…ideas and insights…hobbies and hopes. Most of all, you have important contributions to make as we pursue solutions that will impact your life. That’s why we pledge to bring five key qualities to every patient relationship:

Compassion
We will always listen with
our ears and our hearts.
Empathy
Your struggles are not lost on us
and we know how real they are.
Dedication
We embrace the perseverance
that true progress requires.
Focused Intensity
It’s the only way to do this work.
Commitment
This work is important and we give it our all.
To learn more about the relationships we build with patients, send us a message.
Compassion
We will always listen with
our ears and our hearts.
Empathy
Your struggles are not lost on us
and we know how real they are.
Dedication
We embrace the perseverance
that true progress requires.
Focused Intensity
It’s the only way to do this work.
Commitment
This work is important and we give it our all.
To learn more about the relationships we build with patients, send us a message.
Read the Latest
June 30, 2025
Crinetics to Showcase the Next Generation of Endocrinology Innovation at ENDO 2025 with Eight Presentations From its Deep Pipeline
Crinetics announced eight abstracts from its novel clinical development programs will be presented at the Endocrine Society’s Annual Meeting, ENDO 2025, July 12-15, 2025, in San Francisco, California.
June 16, 2025
Crinetics Pharmaceuticals to Host R&D Day on June 26, 2025
Crinetics Pharmaceuticals will host an in-person and virtual R&D Day in New York on Thursday, June 26, 2025, from 9:00 AM to 12:00 PM ET.
June 10, 2025
Crinetics Pharmaceuticals Announces June 2025 Inducement Grants Under Nasdaq Listing Rule 5635(c)(4)
Crinetics granted non-qualified stock option awards to purchase an aggregate of 74,000 shares of its common stock and granted an aggregate of 48,850 restricted stock unit awards to 20 new non-executive employees.
May 23, 2025
Crinetics Pharmaceuticals to Participate in the Jefferies Global Healthcare Conference 2025
will participate in the Jefferies Global Healthcare Conference, taking place June 3-5, 2025 in New York, NY.






